



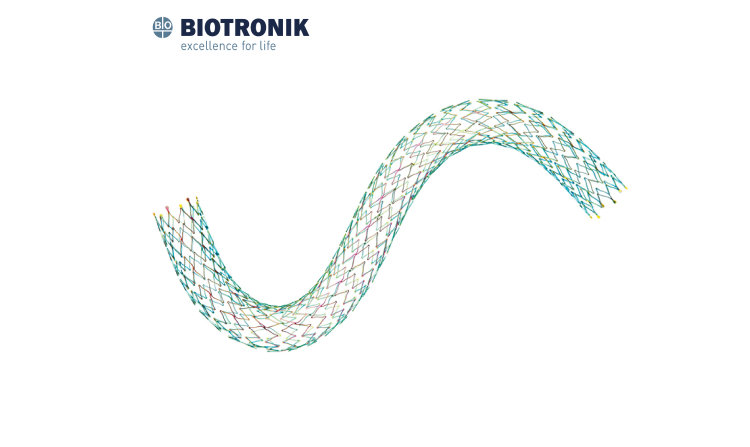
Pulsar-18 T3

Indicated for use in patients with atherosclerotic disease of the superficial femoral, proximal popliteal and infrapopliteal arteries and for the treatment of insufficient results after Percutaneous Transluminal Angioplasty (PTA), e.g. residual stenosis and dissection.*
Technical Data
| Stent | |
|---|---|
| Catheter type | OTW |
| Recommended guide wire | 0.018” |
| Stent material | Nitinol |
| Strut thickness | 140 μm |
| Strut width | 85 μm |
| Stent coating | proBIO® (Amorphous Silicon Carbide) |
| Stent Markers | 6 gold markers each end |
| Sizes | ø 4.0 - 7.0 mm: L:20 - 200 mm |
| Shaft | 4F, hydrophobic coating, tri-axial |
| Usable length | 90 cm and 135 cm |
Ordering Information
| Stent ø (mm) | Catheter length 90 cm (Stent length mm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20** | 30 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | |||||
| 4.0 (4F) | 430437 | 430438 | 430439 | 430440 | 430441 | 430442 | 430443 | 430444 | 430445 | 430446 | ||||
| 5.0 (4F) | 430447 | 430448 | 430449 | 430450 | 430451 | 430452 | 430453 | 430454 | 430455 | 430456 | ||||
| 6.0 (4F) | 430457 | 430458 | 430459 | 430460 | 430461 | 430462 | 430463 | 430464 | 430465 | 430466 | ||||
| 7.0 (4F) | 430467 | 430468 | 430469 | 430470 | 430471 | 430472 | 430473 | 430474 | 430475 | 430476 | ||||
| Stent ø (mm) | Catheter length 135 cm (Stent length mm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20** | 30 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | |||||
| 4.0 (4F) | 430477 | 430478 | 430479 | 430480 | 430481 | 430482 | 430483 | 430484 | 430485 | 430486 | ||||
| 5.0 (4F) | 430487 | 430488 | 430489 | 430490 | 430491 | 430492 | 430493 | 430494 | 430495 | 430496 | ||||
| 6.0 (4F) | 430497 | 430498 | 430499 | 430500 | 430501 | 430502 | 430503 | 430504 | 430505 | 430506 | ||||
| 7.0 (4F) | 430507 | 430508 | 430509 | 430510 | 430511 | 430512 | 430513 | 430514 | 430515 | 430516 | ||||










 Временные кардиостимуляторы
Временные кардиостимуляторы Постоянные кардиостимуляторы
Постоянные кардиостимуляторы Проводы временного кардиостимулятора
Проводы временного кардиостимулятора Домашний мониторинг
Домашний мониторинг Инструменты
Инструменты